PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1693673

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1693673
China Active Pharmaceutical Ingredients (API) - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
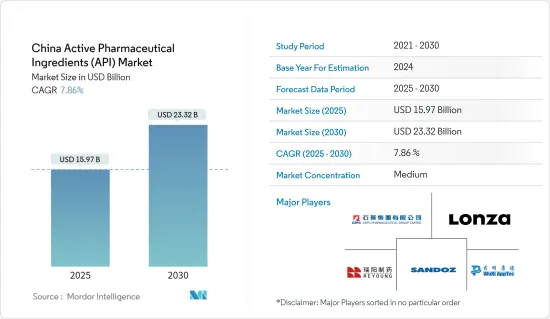
The China Active Pharmaceutical Ingredients Market size is estimated at USD 15.97 billion in 2025, and is expected to reach USD 23.32 billion by 2030, at a CAGR of 7.86% during the forecast period (2025-2030).
The market is expected to grow due to the increasing prevalence of chronic disorders and the rising adoption and approval of biologics and biosimilars during the forecast period.
The increasing prevalence and incidence of chronic diseases, infectious diseases, and genetic disorders in China are driving the demand for effective and safe drugs. Thus, the need for active pharmaceutical ingredients across the country is increasing for the development of novel drugs for various diseases. This, in turn, is further expected to fuel the growth of the active pharmaceutical ingredients market during the forecast period. For instance, according to an article published in the Chinese Medical Journal in March 2022, it has been observed that about 4.82 million new cancer cases were diagnosed in China in 2022, compared to 4.5 million cases in the last year. Thus, the expected increase in the number of people who have cancer, including colorectal cancer, raises the demand for novel drugs for the treatment. This, in turn, is anticipated to propel the growth of the APIs market during the forecast period.
Also, ischemic heart disease, stroke, Alzheimer's and other dementias, and chronic obstructive pulmonary disease are the four most frequent causes of disability-adjusted life years among the population. For instance, according to an article published in BMC Public Health in June 2023, it was observed that about 40 million people living in China are expected to suffer from Alzheimer's disease and related dementias by 2050. Thus, the expected increase in the patient population raises the need to develop novel biological drugs, which is expected to increase the demand for APIs in China.
Moreover, the increasing adoption and approval of biologics and biosimilars are expected to drive the market during the forecast period. Healthcare systems worldwide actively seek ways to lower costs and raise revenues. Innovative formulary management provides a way to save prescription prices without sacrificing patient care. Specialty pharmaceuticals are one prominent area in which cost reductions would make a significant difference because of the high cost of biologics, which results in a disproportionate contribution to the total national drug spending. Thus, using less expensive biosimilars offers a chance to reduce drug costs and spending on biologics without compromising patient outcomes.
For instance, according to an article published in Health Policy in October 2023, it was observed that the cost of cancer biosimilars was 69% to 90% of the costs for the reference drugs, and their uptake reached 54% to 83% in China in 2022. Hence, biosimilars' lower cost and clinical benefits are expected to increase their adoption, ultimately driving the market during the forecast period. Similarly, in an article published in Bioengineered in June 2022, the Chinese researchers observed that biosimilar monoclonal antibodies are one of the hotspots of research and development in China, with continuous improvement of policies and increasing interest in research and development of biosimilars.
Thus, owing to the development of biopharmaceuticals markets, the demand for biosimilars in China is expected to grow gradually, with the improvement of the national guidelines, further increasing its adoption for the treatment of various chronic and infectious disorders, contributing to the market growth.
Therefore, the market is expected to grow during the forecast period due to the aforementioned factors, such as the increasing prevalence of chronic diseases and the increasing adoption and approval of biologics and biosimilars. However, the country's stringent regulations for drug approvals and drug price policies are expected to hinder market growth during the forecast period.
China Active Pharmaceutical Ingredients (API) Market Trends
The Oncology Segment is Expected to Register a High Annual Growth Rate
The growing burden of cancer cases is one of the primary healthcare concerns in China. The incidence of cancer rises dramatically with age, most likely due to a build-up of risks for specific cancers that increase with age. Hence, there should be proper management of cancer, which is possible with advanced medicines. Thus, the growing need for advanced medicines for the management of various types of cancers is projected to spur the demand for APIs to manufacture finished dosage forms of cancer medicine.
The significant burden of cancer cases in China is projected to foster demand for innovative medicines for cancer treatment, which is, in turn, projected to accelerate industry expansion during the forecast period. For instance, according to an article published in BMC Cancer in January 2023, there were 4,16,371 new cases of breast cancer in China over the last three years. Thus, the high prevalence of cancers is expected to drive the demand for new medicines for cancer treatment, which will likely support the country's market growth.
In addition, the increasing geriatric population in the country and a higher burden of cancer in the elderly population are projected to foster the country's market growth during the forecast period. For instance, according to an article published by the Science China Life Sciences Journal in December 2022, the burden of cancer among the geriatric population was higher in China. It is estimated that there were 2.79 million cases in older people, representing 55.8% of the overall population in 2022. Thus, the escalating burden of cancer among the Chinese population is anticipated to create a robust demand for novel drug therapies for cancer management, which, in turn, is projected to spur industry expansion.
Also, the rising company focus on developing various cancer-treating drugs raises the demand for APIs, propelling the market growth. For instance, in December 2023, AstraZeneca acquired Gracell, a Chinese cancer therapy firm, for USD 1.2 billion. The deal marks a further investment in cancer research and treatment in China, accounting for about one-third of AstraZeneca's business and its continued push to expand in China. Similarly, in January 2024, Glenmark Pharmaceuticals partnered with 3D Medicines and Jiangsu Alphamab Biopharmaceuticals to launch envafolimab to treat various types of cancers. Under this agreement, Jiangsu Alphamab is the manufacturer of envafolimab.
Thus, such agreements are projected to spur industry expansion during the forecast period. Hence, increasing strategic activities, such as acquisitions and collaborations by key players, are also expected to drive the segment revenue during the forecast period.
Therefore, owing to the above-mentioned factors, the rising prevalence of cancer cases in China and increasing strategic activities by key players are expected to drive the market during the forecast period.
The Branded Drugs Segment is Expected to Hold Significant Market Share
Branded drugs are usually innovative pharmaceuticals with legal patent protection for a specific period. When any investigational candidate fulfills all the research requirements, it is represented as a brand for a specific disease area. The segment growth is mainly driven by the increasing demand for branded medicines and robust API manufacturing facilities to manufacture the API required for the formulation of branded pharmaceuticals. In addition, the growing pharmaceutical market in the country and various strategic initiatives undertaken by market players are further projected to foster segment expansion during the forecast period.
China is one of the major pharmaceutical markets in the world. Hence, most market players launch and export their products in Chinese and international markets to strengthen their business avenues. Thus, various strategic initiatives undertaken by pharmaceutical companies, such as approvals, new launches, investments, and business expansions, are projected to foster demand for APIs to manufacture branded medicines in the country. This is projected to support segment expansion during the forecast period. For instance, in January 2024, Biogen and Eisai's Leqembi received approval from China's National Medical Products Administration. Leqembi is the antibody-based therapeutics used in the treatment of Alzheimer's disease. China has become the third country to approve Leqembi, following the United States and Japan. Thus, the approval of the new product is expected to propel the demand for Leqembi's API production in the country, which is expected to foster market growth during the forecast period. Similarly, the increased efforts of global pharmaceutical players to invest in the Chinese pharmaceutical market are expected to spur the development of novel therapies, which, in turn, are projected to fuel segment uptake over the study period. For instance, in January 2024, Bayer AG and RTW Investments, LP invested USD 35 million and 127 million in Ji Xing Pharmaceuticals Limited of China. This investment was made to accelerate the development of cardiovascular and ophthalmology medicines in China. In addition, this investment allowed Bayer AG to strengthen its presence in the Chinese market. Thus, such investments are expected to hold a strong promise for the development of branded medicines, which, in turn, is expected to foster the demand for APIs.
The segment is further expected to benefit from several efforts undertaken by market players to foster API production capabilities. For instance, in September 2022, Asymchem agreed to transfer land in Taixing, Jiangsu Province, China, to construct a proposed R&D and production facility for chemical raw materials, small-molecule APIs, and drug products. Thus, such instances are expected to strengthen API manufacturing capabilities in the country, which is expected to spur segment market growth during the forecast period.
Thus, the above-mentioned factors, increasing demand for branded products, growing pharmaceutical markets, and several strategic initiatives undertaken by companies are expected to accelerate segment expansion during the forecast period.
China Active Pharmaceutical Ingredients (API) Industry Overview
The Chinese active pharmaceutical ingredients market is relatively fragmented. The API market has several manufacturers focusing on expanding their footprints by adopting various business strategies, such as collaborations, facility expansion, and drug approvals. Some key market players include CSPC Pharmaceutical Group Limited (Shijiazhuang Pharmaceutical Group Co. Ltd), WuXi AppTec (WuXi STA), and Lonza.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope Of The Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Prevalence of Chronic Disorders
- 4.2.2 Increasing Adoption and Approval of Biologicals and Biosimilars
- 4.3 Market Restraints
- 4.3.1 Drug Price Control Policies
- 4.3.2 High Competition Between API Manufacturers and Stringent Regulations
- 4.4 Industry Attractiveness - Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Business Mode
- 5.1.1 Captive API
- 5.1.2 Merchant API
- 5.2 By Synthesis Type
- 5.2.1 Synthetic
- 5.2.2 Biotech
- 5.3 By Drug Type
- 5.3.1 Generic
- 5.3.2 Branded
- 5.4 By Application
- 5.4.1 Cardiology
- 5.4.2 Oncology
- 5.4.3 Pulmonology
- 5.4.4 Neurology
- 5.4.5 Orthopedic
- 5.4.6 Ophthalmology
- 5.4.7 Other Applications
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Teva Pharmaceutical Industries Ltd
- 6.1.2 Shenzhen Shengda Pharma Limited
- 6.1.3 Jinzhou Pharmaceuticals
- 6.1.4 WuXi AppTec
- 6.1.5 Sandoz Group AG
- 6.1.6 Pfizer Inc.
- 6.1.7 Jinan Shengqi Pharmaceutical Co. Ltd
- 6.1.8 Reyoung Pharmaceuticals
- 6.1.9 Lonza
- 6.1.10 CSPC Pharmaceutical Group Limited (Shijiazhuang Pharmaceutical Group Co. Ltd)
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




