Need help finding what you are looking for?
Contact Us
PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1640649

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1640649
Europe Pharmaceutical Contract Manufacturing - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
PUBLISHED:
PAGES: 105 Pages
DELIVERY TIME: 2-3 business days
SELECT AN OPTION
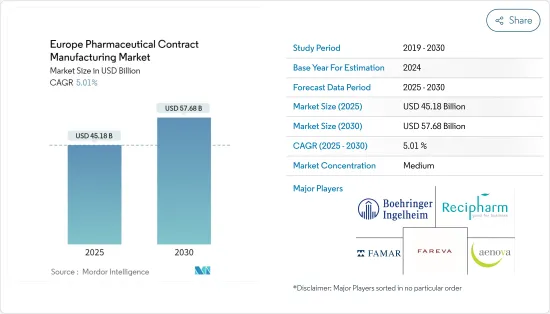
The Europe Pharmaceutical Contract Manufacturing Market size is estimated at USD 45.18 billion in 2025, and is expected to reach USD 57.68 billion by 2030, at a CAGR of 5.01% during the forecast period (2025-2030).
Key Highlights
- By outsourcing production, pharmaceutical companies can focus on core competencies like research and development, marketing, and sales. Contract manufacturers bring expertise, state-of-the-art facilities, and cost-effective operations, allowing pharma companies to accelerate time-to-market, increase production capacity, and reduce operational costs. These benefits are encouraging pharmaceutical firms to opt for contract manufacturing.
- Furthermore, pharmaceutical companies are increasing their R&D expenditures. The level of R&D spending is a crucial indicator of a pharmaceutical company's commitment to drug discovery. Consequently, increased government investments in pharmaceutical R&D are expected to enhance research activities, improve the conversion of products into market-ready forms through pharmaceutical contract manufacturers, and drive the market's growth. For instance, according to the European Federation of Pharmaceutical Industries and Associations (EFPIA) and Pharmaceutical Research and Manufacturers of America (PhRMA), the pharmaceutical research and development spending annual growth rate in Europe registered a growth rate of 6.7% in 2019-2023 increased from 3.7% in 2014-2018.
- However, in Europe, the pharmaceutical industry's preference for maintaining control over manufacturing processes is limiting the market's growth. Despite the cost savings and operational efficiencies that outsourcing offers, companies are reluctant to give up control over critical production aspects. This hesitation is primarily due to concerns about intellectual property protection, quality standards, and supply chain reliability.
- Contract-based manufacturers in the United Kingdom encounter challenges due to the political implications of the United Kingdom's departure from the EU. UK/EU-based CMOs must adhere to rigorous testing protocols for various products under two regulatory frameworks. The government is promoting increased exports to diversify the UK economy away from its traditional reliance on financial services and credit-driven spending. As a result, pharmaceutical companies with limited production capacities may seek to outsource parts of their manufacturing operations, thereby increasing the demand for CMOs.
- The pandemic had a varied impact on the market studied, further driving its growth in the region. This was primarily due to the increased demand for vaccines, therapeutics, and personal protective equipment, which strained manufacturing capacities. As a result, contract manufacturers expanded production to meet the demand. However, supply chain disruptions, labor shortages, and regulatory challenges posed some obstacles.
Europe Pharmaceutical Contract Manufacturing Market Trends
The Active Pharmaceutical Ingredient (API) Segment Holds a Major Share in the Market
- The aging population and the rising prevalence of chronic diseases are driving the increased demand for drugs. Also, the focus on developing generic drugs and biosimilars is boosting the demand for active pharmaceutical ingredients (APIs). Furthermore, ongoing research and development in the pharmaceutical sector, coupled with rising healthcare expenditures, are further driving API demand across European countries.
- In response to the increasing demand for APIs, manufacturers are witnessing investment in the expansion of API manufacturing with more capacity. In June 2024, ESTEVE commenced the construction of a new manufacturing unit at its Celra plant in Girona, with an investment plan of EUR 100 million (USD 14.07 million) by 2026. This expansion aims to enhance the production capacity of active pharmaceutical ingredients (API) at ESTEVE's primary industrial hub. The project includes the construction of new production and service buildings and the installation of reaction volumes totaling up to 150 cubic meters. Consequently, ESTEVE's Celra production capacity will increase by 45%, contributing an additional 15% to the company's global capacity. This development at the Girona plant represents a significant advancement in ESTEVE's business of developing, manufacturing, and selling active ingredients for third-party clients (CMO).
- Companies are actively pursuing acquisition and expansion strategies to bolster API manufacturing, driving growth in the sector. For instance, in January 2023, an active pharmaceutical ingredient (API) production plant in Ringaskiddy, Ireland, was acquired by Sterling Pharma Solutions, a multinational contract development and manufacturing company from Novartis. Additionally, the deal includes an ongoing supply agreement with Novartis to continue to manufacture several APIs for cardiology, immunology, and oncology medications at Ringaskiddy. The facility will add capacity to Sterling's expanding API manufacturing capabilities. Financial information was kept private.
- The increasing revenue from APIs in Europe highlights the significant advancements made by pharmaceutical manufacturers. For instance, in FY2023, Cipla Limited generated approximately 39% of its revenue from APIs in European and global markets, underscoring the company's strategic focus on this segment.
Germany is Projected to Drive the Market's Growth Rapidly
- Germany is likely to maintain its position as the premier pharmaceutical contract manufacturing market in Western Europe, both in capacity and market share. The region attracts pharmaceutical manufacturers due to its highly skilled and specialized workforce, which is essential for producing highly potent active pharmaceutical ingredients (HPAPIs).
- The country is renowned for its strong intellectual property protections and effective measures against organized crime. The steady annual growth rate is primarily due to the well-established expertise of German CMOs and the government's supportive policies for generics. Approximately 60% of Germany's pharmaceutical products are exported to major markets, including the United States, the United Kingdom, Belgium, the Netherlands, and Switzerland. Furthermore, high productivity levels and a competitive tax system significantly drive the pharmaceutical CMO market in Germany.
- Moreover, as part of their growth strategy, companies are increasingly targeting new regions. For instance, in October 2023, Vetter Pharma, a contract development and manufacturing organization, announced an investment of USD 243 million in a new production facility at its global headquarters in Ravensburg, Germany. This construction aims to enhance aseptic manufacturing capacity and introduce several new commercial filling lines. The initial cleanrooms are scheduled for installation by the end of 2024. Beyond the production facility, the investment includes expanding lab space for analytical services and increasing filling capacity with new commercial production lines at other EU locations.
- Leading German manufacturers and service providers, including BASF, B. Braun, Evonik, Vetter Pharma AG, Wacker, Corden Pharma, and Merck, are significantly enhancing CMO activities in the country's pharmaceutical sector, contributing to the market's growth during the forecast period.
Europe Pharmaceutical Contract Manufacturing Industry Overview
The European pharmaceutical contract manufacturing market is consolidated and consists of a few players. In terms of market share, top companies have control over the market. Major players include Fareva Holdings SA, Recipharm AB, Boehringer Ingelheim Group, Aenova Group, Famar SA, and Lonza Group.
- May 2024: EUROAPI, a pharmaceutical company, signed a CMO agreement with a global animal health company. Under this partnership, EUROAPI supplies veterinary products to its clients. The contract's total value is estimated to be between EUR 130 million and EUR 150 million from 2025 to 2029.
- November 2023: NorthEdge invested in Torbay Pharmaceuticals (Torbay), a contract manufacturer and license holder specializing in sterile injectables, from Torbay and Devon NHS Foundation Trust. This investment aims to support Torbay's next phase of international expansion. Torbay has experienced rapid growth, driven by increasing demand for commercial-scale sterile injectable fill/finish services. The company supplies healthcare organizations and pharmaceutical firms in the United Kingdom and internationally.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
Product Code: 56254
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET INSIGHTS
- 4.1 Market Overview
- 4.2 Industry Attractiveness - Porter's Five Forces Analysis
- 4.2.1 Bargaining Power of Suppliers
- 4.2.2 Bargaining Power of Consumers
- 4.2.3 Threat of New Entrants
- 4.2.4 Intensity of Competitive Rivalry
- 4.2.5 Threat of Substitutes
- 4.3 Industry Value Chain Analysis
- 4.4 Industry Policies
- 4.5 Assessment of the Impact of Macro-economic Factors on the Market
5 MARKET INSIGHTS
- 5.1 Market Drivers
- 5.1.1 Increasing Outsourcing Volume by Pharmaceutical Companies
- 5.1.2 Increasing Investment in R&D
- 5.2 Market Restraints
- 5.2.1 Increasing Lead Time and Logistics Costs
- 5.2.2 Stringent Regulatory Requirements
- 5.2.3 Capacity Utilization Issues Affecting the Profitability of CMOs
6 TECHNOLOGY SNAPSHOT
7 MARKET SEGMENTATION
- 7.1 By Service Type
- 7.1.1 Active Pharmaceutical Ingredient (API) Manufacturing
- 7.1.2 Finished Dosage Formulation (FDF) Development and Manufacturing
- 7.1.2.1 Solid Dose Formulation
- 7.1.2.2 Liquid Dose Formulation
- 7.1.2.3 Injectable Dose Formulation
- 7.1.3 Secondary Packaging
- 7.2 By Country
- 7.2.1 United Kingdom
- 7.2.2 Germany
- 7.2.3 France
- 7.2.4 Italy
- 7.2.5 Rest of Europe
8 COMPETITIVE LANDSCAPE
- 8.1 Company Profiles
- 8.1.1 Fareva Holdings SA
- 8.1.2 Recipharm AB
- 8.1.3 Boehringer Ingelheim Group
- 8.1.4 Aenova Group
- 8.1.5 Famar SA
- 8.1.6 Lonza Group
- 8.1.7 Cenexi - Laboratoires Thissen SA
- 8.1.8 Almac Group
9 INVESTMENT ANALYSIS
10 FUTURE OF THE MARKET
Have a question?


SELECT AN OPTION
Have a question?


Questions? Please give us a call or visit the contact form.
