PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1708858

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1708858
Global Allan-Herndon-Dudley Syndrome Treatment Market, By Treatment Type (Gene Therapy, Hormone Therapy), By End User (Hospital, Specialty Clinics, Others), By Geography (North America, Europe, Asia Pacific, Latin America, Middle East and Africa)
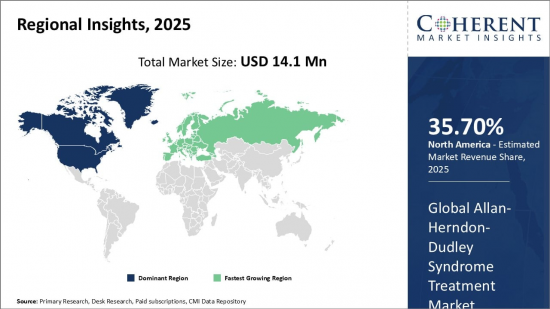
Global Allan-Herndon-Dudley Syndrome Treatment Market is estimated to be valued at USD 14.1 Mn in 2025 and is expected to reach USD 20.8 Mn by 2032, growing at a compound annual growth rate (CAGR) of 5.7% from 2025 to 2032.
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 14.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 5.70% | 2032 Value Projection: | USD 20.8 Mn |
Allan-Herndon-Dudley syndrome (AHDS) is a rare genetic disorder caused by mutations in the MCT8 gene. These mutations result in impaired thyroid hormone transport across the blood-brain barrier. Current treatment options for AHDS are limited and aim to manage symptoms. The first line of treatment involves thyroid hormone replacement therapy with triiodothyronine (T3). T3 supplements help restore thyroid hormone levels in the brain to improve neurological development and function. However, high T3 doses sometimes cause side effects like tremors, hyperactivity, and irregular heartbeat. The other option is thyroxine (T4) therapy which relies on the peripheral conversion of T4 to active T3 within cells. This indirect method of treatment is not very effective as the MCT8 mutations disrupt hormone transportation and activation.
Emerging gene therapy is a promising investigational approach to directly treat the underlying genetic cause of AHDS. In gene therapy, a normal copy of the MCT8 gene would be delivered into the patient's cells and tissues to replace the mutated one. Though still in early research phase, this could correct the hormone transport defect and eliminate the need for lifelong thyroid supplementation. Challenges include developing effective and safe gene delivery vectors and treatments needing to be tailored to individual patients based on their specific MCT8 mutations. More research is underway to validate gene therapy as an innovative treatment for AHDS in the future.
Market Dynamics:
The global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) treatment market is mainly driven by the increasing prevalence of AHDS is expected to propel the market growth over the forecast period. For instance, according to the data published by the National Center for Biotechnology Information (NCBI) in January 16, 2020, the identification of more than 160 affected individuals in approximately 15 years suggests that the syndrome is more common than previously thought.
Key features of the study:
- This report provides an in-depth analysis of the global allan-herndon-dudley syndrome (AHDS) (MCT8 deficiency) treatment market, and provides market size (US$ Mn) and compound annual growth rate (CAGR %) for the forecast period (2025-2032), considering 2024 as the base year
- It elucidates potential revenue growth opportunities across different segments and explains attractive investment proposition matrices for this market
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approvals, market trends, regional outlook, and competitive strategies adopted by key players
- It profiles key players in the global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) treatment market based on the following parameters - company highlights, products portfolio, key highlights, financial performance, and strategies
- Key companies covered as a part of this study include Rare Thyroid Therapeutics International AB and Erasmus Medical Center
- Insights from this report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics
- The global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) treatment market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) treatment market
Detailed Segmentation:
- Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By Treatment Type
- Gene Therapy
- Hormone Therapy
- Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By End User
- Hospital
- Specialty Clinics
- Others (Research institutes, etc.)
- Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By Region
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- Latin America
- Company Profiles
- Rare Thyroid Therapeutics International AB
- Erasmus Medical Center
Table of Contents
1. Research Objectives and Assumptions
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Market Snippet, By Treatment Type
- Market Snippet, By End User
- Market Snippet, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Increasing prevalence of AHDS
- Increasing Healthcare Expenditure Globally
- Key Highlights
- Regulatory Scenario
- Recent Trends
- PEST Analysis
- PORTER's Analysis
- Mergers, Acquisitions, and Collaborations
4. Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market - Impact of Coronavirus (COVID-19) Pandemic
- COVID-19 Epidemiology
- Supply Side and Demand Side Analysis
- Economic Impact
5. Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By Treatment Type, 2020-2032, (US$ Mn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Gene Therapy
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032,(US$ Mn)
- Hormone Therapy
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032,(US$ Mn)
6. Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By End User, 2020-2032, (US$ Mn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Hospital
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032,(US$ Mn)
- Specialty Clinics
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032,(US$ Mn)
- Others (Research institutes, etc.)
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032,(US$ Mn)
7. Global Allan-Herndon-Dudley syndrome (AHDS) (MCT8 deficiency) Treatment Market, By Region, 2020-2032, (US$ Mn)
- Introduction
- Market Share Analysis, By Region, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, For Region, 2021 - 2032
- Country Trends
- North America
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032,(US$ Mn)
- U.S.
- Canada
- Europe
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032,(US$ Mn)
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032,(US$ Mn)
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Latin America
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032,(US$ Mn)
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032,(US$ Mn)
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Treatment Type, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032,(US$ Mn)
- Market Size and Forecast, and Y-o-Y Growth, By Region/Country, 2020-2032,(US$ Mn)
- North Africa
- Central Africa
- South Africa
8. Competitive Landscape
- Company Profile
- Rare Thyroid Therapeutics International AB
- Company Highlights
- Product Portfolio
- Key Highlights
- Financial Performance
- Strategies
- Erasmus Medical Center
- Company Highlights
- Product Portfolio
- Key Highlights
- Financial Performance
- Strategies
- Analyst Views
9. Section
- References
- Research Methodology
- About us




